Development of a liquid crystalline gel based on Amazonian butter extracted from ucuúba seeds (Virola surinamensis (Rol. ex Robbt.) Warb.) for vaginosis treatment
DOI:
https://doi.org/10.70151/nvd9bw73Keywords:
Liquid crystalline gel, Amazonian fat, Sustained drug release, Virola surinamensis, DD solverAbstract
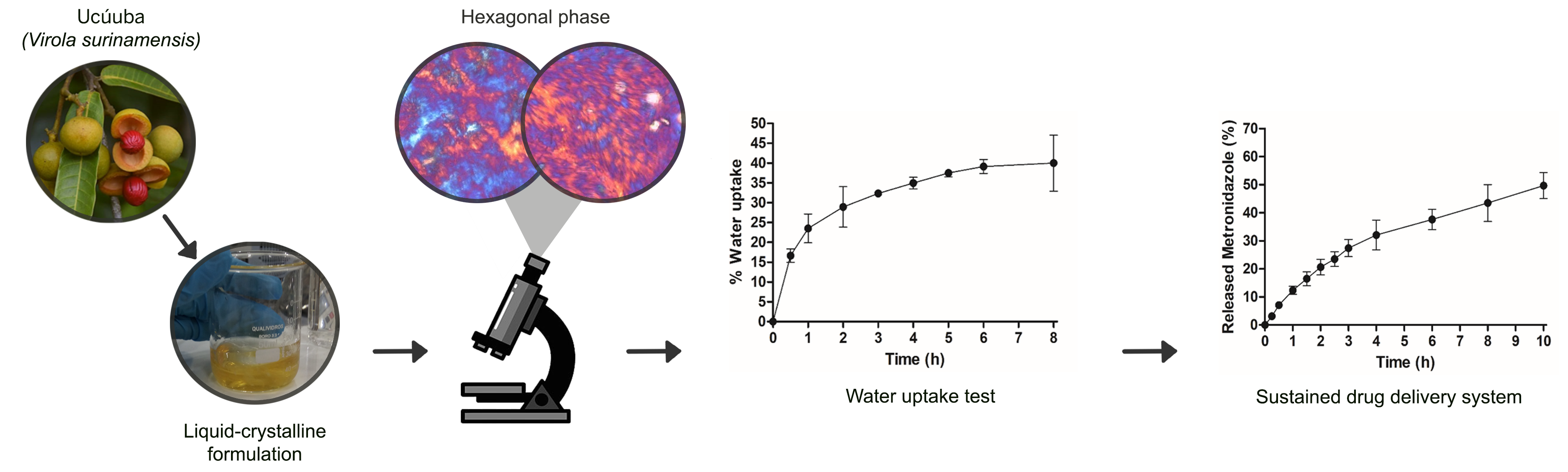
This study focuses on developing a liquid crystalline gel based on ucuúba butter from the Amazon. This gel, which offers distinct technological advantages for vaginal sustained drug release systems, was characterized by polarized light microscopy (PLM), pH measurements, water uptake assay, drug release profile and kinetic study (using the Excel Add-in and DDSolver). The unloaded and drug-loaded liquid crystalline gel containing 10% metronidazole exhibited a hexagonal mesophase structurally favorable for sustained drug release, with a pH value of 6,4 ± 0.024. The water uptake profile showed a fast water uptake in up to 1 h, approximately 16%, with the maximum amount of water, 40%, in 8 h. After water uptake, the liquid crystalline gel undergoes self-assembly to a viscous cubic phase, avoiding rapid erosion in an aqueous medium. In vitro, the release of metronidazole was slow, with 52% of the drug released in 10 h. The best fit was the Higuchi kinetic model with the highest MSC (3,94) and R2 (0,98) values, indicating that the drug release was diffusion-controlled. Korsmeyer’s model showed that formulation exhibited anomalous transport (non-Fickian). Given the limited number of options for topical vaginal treatment, this liquid crystalline gel based on Virola surinamensis (Rol. ex Robbt.) Warb.) butter could potentially find applicability to reduce the frequency of administration and improve local concentrations of metronidazole on vaginal mucous.
Downloads
References
Abraham A, Chan E, Park H (2020) Fast understanding of phases and phase separation in liquid crystal drug delivery systems using deuterium solid-state NMR. Solid State Nucl Magn Reson 106:101642. https://doi.org/10.1016/j.ssnmr.2020.101642
Ahmed AR, Dashevsky A, Bodmeier R (2010) Drug release from and sterilization of in situ cubic phase forming monoglyceride drug delivery systems. Eur J Pharm Biopharm 75(3):375-80. https://doi.org/10.1016/j.ejpb.2010.04.004.
Américo ÁVLS, Nunes KM, Assis FFV, Dias SR, Passos CTS, Morini, AC, Araújo JA, Castro KCF, Silva SKR, Barata LES, Minervino AHH (2020) Efficacy of Phytopharmaceuticals from the Amazonian Plant Libidibia ferrea for Wound Healing in Dogs. Front Vet Sci 12:7:244. https://doi.org/10.3389/fvets.2020.00244
Antunes TC, Salamoni SP, Frazzon APG, Germani JC, Van Der Sand ST (2013) Influence of nutritional source on optimal growth and production of antimicrobials produced by isolates of Streptomyces sp. Braz J Biosc 11(2):131-138.
Bansal M, Mittal N, Yadav SK, Khan G, Gupta P, Mishra B, Nath G (2018) Periodontal thermoresponsive, mucoadhesive dual antimicrobial loaded in-situ gel for the treatment of periodontal disease: Preparation, in-vitro characterization and antimicrobial study. J Oral Biol Craniofac Res 8:126–133. https://doi.org/10.1016/j.jobcr.2017.12.005
Brazil. Ministry of Health. National Health Surveillance Agency (ANVISA). Resolution of the Collegiate Board – RDC No. 166, of July 24, 2017, provides for Validation of Analytical Methods. Official Gazette of the Union, Executive Branch, 25 de jul. 2017. Access at:<https://antigo.anvisa.gov.br/documents/10181/2721567/RDC_166_2017_COMP.pdf/d5fb92b3-6c6b-4130-8670-4e3263763401> Accessed on: march 28, 2023.
Boyd BJ, Whittaker DV, Khoo SM, Davey G. L (2006) Lyotropic liquid crystalline phases formed from glycerate surfactants as sustained release drug delivery systems. Int J Pharm 309:218–226. https://doi.org/10.1016/j.ijpharm.2005.11.033
Calixto GMF, Victorelli FD, Dovigo LN, Chorilli M (2018) Polyethyleneimine and chitosan polymer-based mucoadhesive liquid crystalline systems intended for buccal drug delivery. AAPS PharmSciTech 19:820–836. https://doi.org/10.1208/s12249-017-0890-2
Carvalho FC, Campos ML, Peccinini RG, Gremião MP (2013) Nasal administration of liquid crystal precursor mucoadhesive vehicle as an alternative antiretroviral therapy. Eur J Pharm Biopharm 84(1):219-27. https://doi.org/10.1016/j.ejpb.2012.11.021
Chen Y, Ma P, Gui S (2014) Cubic and hexagonal liquid crystals as drug delivery systems. Biomed Res Int 2014:815981. https://doi.org/10.1155/2014/815981
Chen Y, Liang X, Ma P, Tao Y, Wu X, Wu X, Chu X, Gui S (2015) Phytantriol-based in situ liquid crystals with long-term release for intra-articular administration. AAPS PharmSciTech. 16(4):846-54. https://doi.org/10.1208/s12249-014-0277-6
Da Silva CSM, De Araújo JA, Silveira TS, Castro KCF, Baratto LC, Kaminski RCK, Santos GB, Nunes KM (2021) Wound healing activity of topical formulations containing Mauritia flexuosa oil. Braz J Pharmacogn 31:225–231. https://doi.org/10.1007/s43450-021-00149-2
Feitosa JM, Silva TSA, Xavier AEF, Rodrigues WCS, Silva ACE, Corrêa CVP, Aguiar FS, Mourão RHV, Oliveira EG, Nunes KM (2021) Evaluation of the quality of Amazonian butters as sustainable raw materials for applications in bioproducts. Rev Ciênc Farm Básica Apl - RCFBA 42:e708. https://doi.org/10.4322/2179-443x.0708
Ghate VM, Lewis SA, Prabhu P, Dubey A, Patel N (2016) Nanostructured lipid carriers for the topical delivery of tretinoin. Eur J Pharm Biopharm 108:253–261. https://doi.org/10.1016/j.ejpb.2016.07.026
Jodar A, Soto L, Turell E, Cortés J (2023) Monotherapy With a Non-Hormonal Centella Asiatica, Hyaluronic Acid, and Prebiotic-Based Vaginal Gel in Women With Bacterial Vaginosis: Case Series. Clin Med Insights Case Rep 23:16:117954762311572. https://doi.org/10.1177/11795476231157244
Kaambo E, Africa C, Chambuso R, Passmore JAS (2018) Vaginal Microbiomes Associated with Aerobic Vaginitis and Bacterial Vaginosis. Front Public Health 6:78. https://doi.org/10.3389/fpubh.2018.00078
Karavana SY, Rençbe S, Şenyiğit ZA, Baloğlu E (2012) A New In-Situ Gel Formulation of Itraconazole for Vaginal Administration. Pharmacology & Pharmacy, 03(04):417–426. https://doi.org/10.4236/pp.2012.34056
Karnwal A, Kumar G, Pant G, Hossain K, Ahmad A, Alshammari MB (2023) Perspectives on Usage of Functional Nanomaterials in Antimicrobial Therapy for Antibiotic-Resistant Bacterial Infections. ACS Omega. https://doi.org/10.1021/acsomega.3c00110
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA (1983) Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm 15(1):25–35. https://doi.org/10.1016/0378-5173(83)90064-9
Kulkarni CV, Wachter W, Iglesias-Salto G, Engelskirchen S, Ahualli S (2011) Monoolein: a magic lipid? Phys. Chem. Chem. Phys 13:3004–3021. https://doi.org/10.1039/C0CP01539C
Lara MG, Bentley MV, Collett JH (2005) In vitro drug release mechanism and drug loading studies of cubic phase gels. Int J Pharm 293:241–250. https://doi.org/10.1016/j.ijpharm.2005.01.008
Lee WN, Salleh NM, Cheng SF (2021) Synthesis and characterization of new Schiff base ester liquid crystals with fatty acids from palm oil as flexible alkyl chain. Ind Crops Prod 170:113808. https://doi.org/10.1016/j.indcrop.2021.113808
Nunes KM, Teixeira CCC, Kaminski RCK, Sarmento VHV, Couto RO, Pulcinelli SH, Freitas O (2016) The monoglyceride content affects the self-assembly behavior, rheological properties, syringeability, and mucoadhesion of in situ–gelling liquid crystalline phase. J Pharm Sci 105:2355–2364. https://doi.org/10.1016/j.xphs.2016.05.005
Pedreiro LN, Cury BSF, Chaud MV, Gremião MPD (2016) A novel approach in mucoadhesive drug delivery system to improve zidovudine intestinal permeability. Brazilian J Pharm Sci 52:715–725. https://doi.org/10.1590/s1984-82502016000400016
Peppas NA (1985) Analysis of Fickian and non-Fickian drug release from polymers. Pharma Acta Helv 60:4:110–111.
Perioli L, Pagano C, Mazzitelli S, Rossi C, Nastruzzi C (2008) Rheological and functional characterization of new antiinflammatory delivery systems designed for buccal administration. Int J Pharm 356:19-28. https://doi.org/10.1016/j.ijpharm.2007.12.027
Petrilli R (2013) Hexagonal liquid crystalline nanoparticles functionalized with transduction peptides for siRNA delivery in the therapy of topical diseases. 55p. Dissertation, Faculty of Pharmaceutical Sciences, Universidade de São Paulo, Ribeirão Preto, Brazil.
Pozharani LB, Baloglu E, Suer K, Guler E, Burgaz EV, Kunter I (2023) Development and optimization of in-situ gels for vaginal delivery of metronidazole and curcumin via box-behnken design: In vitro characterization and anti-trichomonas activity. J Drug Deliv Sci Technol 86:104739. https://doi.org/10.1016/j.jddst.2023.104739
Ritger PL, Peppas NA (1987) A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J Control Release 5(1):37–42. https://doi.org/10.1016/0168-3659(87)90035-6
Sharma R, Kumar S, Malviya R, Prajapati BG, Puri D, Limmatvapirat S, Sriamornsak P (2024) Recent advances in biopolymer-based mucoadhesive drug delivery systems for oral application. J Drug Deliv Sci Technol 91:105227 https://doi.org/10.1016/j.jddst.2023.105227
Superti F, De Seta F (2020) Warding Off Recurrent Yeast and Bacterial Vaginal Infections: Lactoferrin and Lactobacilli. Microorganisms 8(1):130. https://doi.org/10.3390/microorganisms8010130
Wu X, Cheng D, Lu Y, Rong R, Kong Y, Wang X, Niu B (2023) A liquid crystal in situ gel based on rotigotine for the treatment of Parkinson’s disease. Drug Deliv Transl Re 14:1048-1062. https://www.doi.org/0.1007/s13346-023-01449-x
Zhang Y, Huo M, Zhou J, Zou A, Li W, Yao C, Xie S (2010) DDSolver: An Add-In Program for Modeling and Comparison of Drug Dissolution Profiles. AAPS J 12(3):263–271. https://doi.org/10.1208/s12248-010-9185-1
Zuo J, Gao Y, Bou-Chacra N, Löbenberg R (2014) Evaluation of the DDSolver Software Applications. Biomed Res Int 2014:1–9. https://doi.org/10.1155/2014/204925
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

